Bacterial Endotoxins LAL Testing Sop is based on End Point Chromogenic Method.
{getToc} $title={Table of Contents}
1. Purpose:-
It is established to provide a procedure for the bacterial endotoxins testing in materials & products by using endpoint chromogenic method.
2. Scope:-
This procedure is applicable to those materials & products for which endotoxin testing is required.
3. Responsibilities:-
- Quality Control Manager
- Microbiologist
- Sr. Officer QC
4. Definitions:-
Bacterial Endotoxins: Pyrogens are fever-producing materials that most often originate from gram-negative bacterial cell walls, but can also originate as leachates from some chemicals and materials. Pyrogens from bacterial cell walls (the most commonly encountered type of pyrogen) are referred to as bacterial endotoxin and are readily detected by Limulus Amebocyte Lysate (LAL) testing systems.
5. Flow Chart:
6. Description / Procedure:
Toxicolor Kit Components:
6.1 Endotoxins Standard
6.1.1 One vial of endotoxin standard is provided in the kit having potency 0.5EU/ml.
6.1.2 One vial of 2.8ml of LAL Reagent Water is (LRW) also included in the kit for the reconstitution of standard.
6.1.3 Reconstitute the endotoxin standard with 2.8ml of LRW for the preparation of 0.19EU/ml of endotoxin standard.
6.1.4 Store reconstituted endotoxin solution at 2-8˚C in a refrigerator and don’t use this solution after 20% decline in absorbance compared with the absorbance on the first day of reconstitution.
6.2 Toxicolor Lysate (ES 24S or 48S)
6.2.1 Reconstitute the test tube containing lysate with 200µl of the buffer provided in the kit. This lysate is sufficient for four tests as 50µl is used per test.
6.2.2 Quickly stir to dissolve. Avoid air bubbling or fuming during stirring.
6.2.3 Be sure that the reagent is completely dissolved. This reagent should be reconstituted just before use, however, storage of reconstituted lysate must be at -20 to -25˚C in 50ul dispensed quantities in small test tubes. Use stored lysate if the color is not changed from white to yellowish.
6.3 Diazo Coupling Reagent
6.3.1 Four vials are provided in the kit by the manufacturer named as 7s, 8s, 9s & 10s.
6.3.2 Transfer whole contents of the vial 7s to vial 8s.
6.3.3 Add 12ml of distilled water in to remaining vials number 9s & 10s.
6.3.4 So we come with three vials named as 8s, 9s & 10s.
6.3.5 Store reconstituted vials at 2-8˚C in a refrigerator
6.4 TEST PROCEDURE
6.4.1 Adjust pH of the sample in between 5.0 – 7.0 with Pyrogen free 0.1 N NaOH or 0.1 N HCl.
6.4.2 In case of dry powder injections, make a 0.1% solution (50mg in 50ml Pyrogen free water). Fresh water from distillation plant can be used for this purpose.
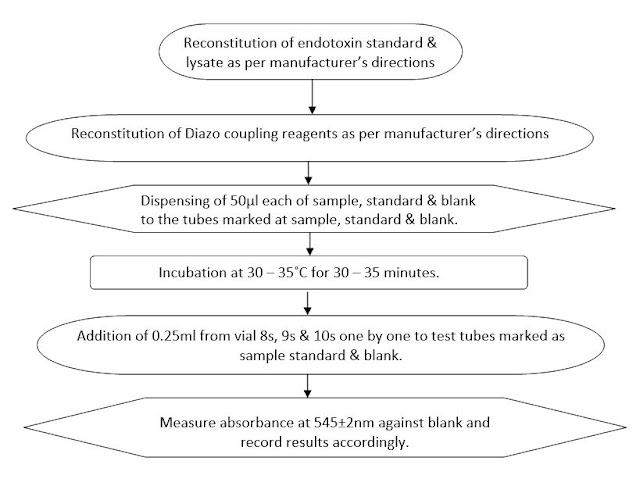
6.4.3 Mark the tubes as sample, standard & blank.
6.4.4 Dispense 50ul test sample in tube marked as sample, 50ul Pyrogen free water used during dilution/solution preparation & 50ul of endotoxin standard in tube marked as standard.
6.4.5 Place the tube stand containing tubes of sample, blank & standard in ice water bath for 5.0 minutes.
6.4.6 Add 50ul lysate into each tube of sample, blank & standard.
6.4.7 Stir the contents of every tube soon after the addition of lysate for few seconds.
6.4.8 Place the test tube rack in incubator at 30 – 35˚C for 30 – 35 minutes.
6.4.9 After the completion of incubation period place the test tube stand in ice water bath tray and add 0.25ml of diazo coupling reagents one by one into each test tube from vial 8s, 9s & 10s. Separate pipette must be used for dispensing of each solution.
6.4.10 Measure absorbance of the test sample and standard against blank at 545±2nm.
Calculate endotoxins contents in the sample according to the following formula:
7. Testing Frequency:-
Every third batch of each product or as and when required in addition to bacterial endotoxin test by gel clot method.
8. Record:-
LAL TEST REPORT (End Point Chromogenic Method)
9. References:-
Manufacturer’s Manual
{getButton} $text={Download in Microsoft Office} $icon={Download} $color={Hex Color}



.webp)






.webp)

